» Back to list of all Management Briefs

| Issue 97 | June 2015 |
Comparative Effectiveness of Proton Irradiation TreatmentMaximizing target tumor dose while minimizing healthy tissue damage continues to be a challenge in radiation therapy. Proton beam therapy (PBT) has held the clinical promise of allowing for higher doses of radiation to be delivered more safely, especially for ocular, skull base, and spinal tumors that require precise control of the exit dose. But the role of protons is less clear for more common tumors, like prostate, where the dosimetric advantages may be diminished and for which intensity-modulated radiation therapy (IMRT) can now safely deliver precise, optimally high radiation doses. Another consideration for PBT is that control of its dose delivery to the tumor and surrounding tissue may be more influenced by tissue alterations due to organ motion, tumor volume change, etc., than with other external beam irradiation therapies. To help consider the increased number of offers from University Affiliates to provide contracted off-site proton irradiation therapy, the VA Radiation Oncology Program requested that the Evidence-based Synthesis Program Coordinating Center (ESP CC) synthesize the most recent literature on the comparative effectiveness of PBT in various cancers. The report of that synthesis focused on the following questions:
Results: For the cancer sites and types reviewed by the ESP report, there are no reliable data from long-term randomized trials on survival, quality of life, or functional capacity of patients who underwent PBT compared with any other modality. The authors could not fully assess the overall net health benefit of proton beam therapy versus its comparators because comparative observational studies did not consistently report many outcomes of greatest interest. Existing comparative studies have numerous methodological deficiencies that limited confidence in the findings, and the findings may have limited applicability across all US proton beam facilities. Although numerous randomized controlled trials are underway that carry the promise of improved toxicity measurement, the report noted it is unclear whether these will fully address gaps in evidence on such outcomes as recurrence, ability to deliver planned chemotherapy and radiation regimens, functional capacity, overall severe late toxicity, and secondary malignancies. Although the report found comparative studies in giant cell tumors, head and neck cancer, uveal hemangiomas, and meningiomas, they provided insufficient evidence for drawing conclusions. There is insufficient evidence to draw conclusions about the comparative effects of PBT versus other radiation modalities among patients with recurrent tumors or how the comparative effects of proton and photon beam therapies differ according to variation in tumor motion. Implications: Despite the common claim that the advantage of proton beam therapy is self-evident, the report authors found no comparative studies demonstrating any common clinical situations in which proton beam therapy has an important clinical advantage with meaningful long-term health outcomes (for example, greater tumor response or reduced radiation side-effects). At the same time, these studies have uncovered low-strength evidence of potential disadvantages of proton beam therapy compared with IMRT and 3D-CRT for breast, esophageal, prostate, and spinal cord glioma cancers. The report's Table 2 (see below) summarizes the key findings on comparative benefits and harms that are supported by low-strength evidence. Because this is still a rapidly evolving field, with ongoing efforts to improve techniques and reduce costs, the authors note their review may need frequent updating to keep up-to-date with emerging research. Reference: Peterson K, McCleery E, Waldrip K. Comparative effectiveness of proton irradiation treatment. VA ESP Project #09-199; 2015. To view the full report, go to http://vaww.hsrd.research.va.gov/publications/esp/ProtonBeam.cfm (Intranet only). 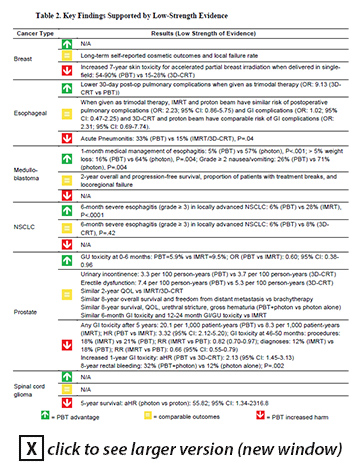
|
Please feel free to forward this information to others! This Management eBrief is a product of the HSR&D Evidence Synthesis Program (ESP). ESP is currently soliciting review topics from the broader VA community. Nominations will be accepted electronically using the online Topic Submission Form. If your topic is selected for a synthesis, you will be contacted by an ESP Center to refine the questions and determine a timeline for the report. This Management e-Brief is provided to inform you about recent HSR&D findings that may be of interest. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs. If you have any questions or comments about this Brief, please email CIDER. The Center for Information Dissemination and Education Resources (CIDER) is a VA HSR&D Resource Center charged with disseminating important HSR&D findings and information to policy makers, managers, clinicians, and researchers working to improve the health and care of Veterans. |
|
- This report is a product of VA/HSR&D's Quality Enhancement Research Initiative's (QUERI) Evidence-Based Synthesis Program (ESP), which was established to provide timely and accurate synthesis of targeted healthcare topics of particular importance to VA managers and policymakers – and to disseminate these reports throughout VA. See all reports online. |
|